What Is the Atomic Number of Iron 58
The periodic table is the tabular arrangement of all the chemical elements on the basis of their respective atomic numbers. In the periodic table, the vertical columns are called 'groups' and the horizontal rows are called 'periods'. The modern periodic table is based on the modern periodic law put forward by the English physicist Henry Moseley, which states that "the properties of elements are periodic functions of their atomic numbers". Periodic trends in the properties of the elements can be observed down the groups and across the periods of the modern periodic table.
Table of Content
- Elements on the periodic table
- Related Videos on Periodic Table of Elements
- List of Chemical Elements
- Atomic Number of Elements
- FAQs
Elements on The Periodic Table
Every chemical element has a specific atomic number, which provides insight into the number of protons present within its nucleus. All isotopes of an element fall under a single cell on the periodic table, since they all share the same atomic number.
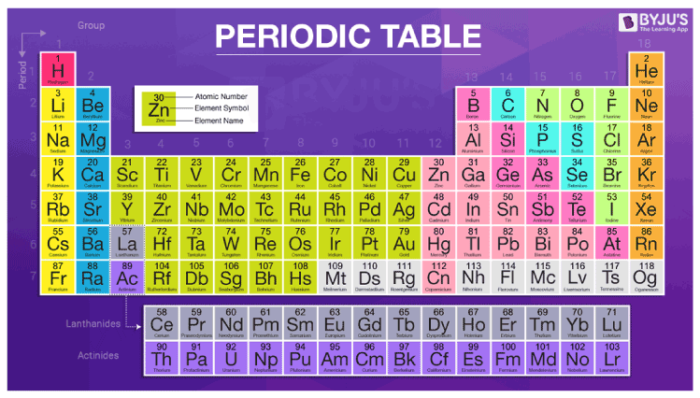
Periodic Table of Elements – Atomic Number, Atomic Mass, Groups & Symbols
Also, Check ⇒
- Periodic Trends in Atomic Radii
- Periodic Trends in Ionic Radii
- Periodic Trends in Electronegativity
- Periodic Trends in Electron Affinity
- Periodic Trends in Ionization Energy

List of Chemical Elements
The table below consists of 118 elements of the periodic table, sorted by atomic number, atomic weight, symbols, density, discovered year and the group.
Atomic Number of Elements
There are about ninety elements found on Earth. Each one has a different number of protons, electrons and neutrons. The total number these subatomic particles held by the respective elements are responsible for their unique properties (including radioactivity).
The number of protons in the nucleus is called the atomic number. The atomic number of each element is unique. The combined number of protons and neutrons in an atom is called the atomic mass number. While the atomic number always stays the same some elements have atoms with different atomic mass numbers. This is because some elements have a different number of neutrons in the nucleus. Versions of an element with different atomic mass numbers are called isotopes.
Frequently Asked Questions – FAQs
What is atomic number?
The atomic number of an element is equal to the total number of protons in the nucleus of the atoms of that element. The atomic number can provide insight into the electronic configuration of the element. For example, carbon has an electron configuration of [He] 2s2 2p2, since its atomic number is 6.
What is the atomic number and mass number?
The number of protons and the number of neutrons shall determine the mass number of an element. Since the isotopes of an element have slightly different mass numbers, it calculates the atomic mass by obtaining the mean of the mass numbers for its isotopes.
Can two different elements have the same atomic number?
Atoms from two different elements may have the same neutron count, but never the same proton count. The number of protons is unique to the element and it represents the number of atoms.
How do we calculate atomic mass?
Add the mass of protons and neutrons to compute the atomic mass of a single atom of an element. Example: Find the atomic mass of a carbon isotope which has 7 neutrons. From the periodic table you can see that carbon has an atomic number of 6, which is its proton number.
Why is atomic number important?
Atomic number is called the number of protons in an atom. This number is very important, because it is unique to a given element's atoms. An element's atoms all have the same number of protons and each element has a different number of protons in its atoms.
What Is the Atomic Number of Iron 58
Source: https://byjus.com/chemistry/periodic-table-elements/
0 Response to "What Is the Atomic Number of Iron 58"
Post a Comment